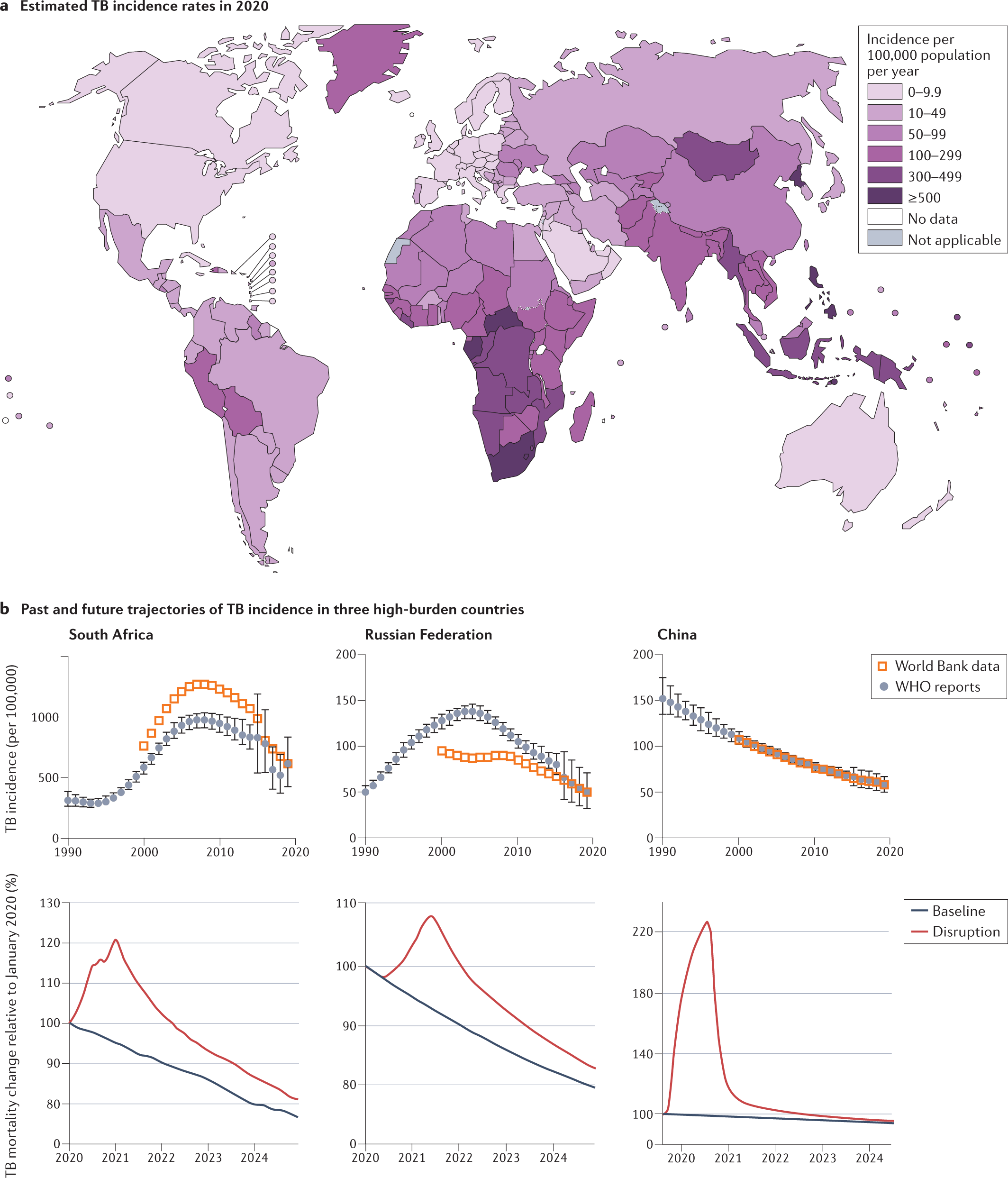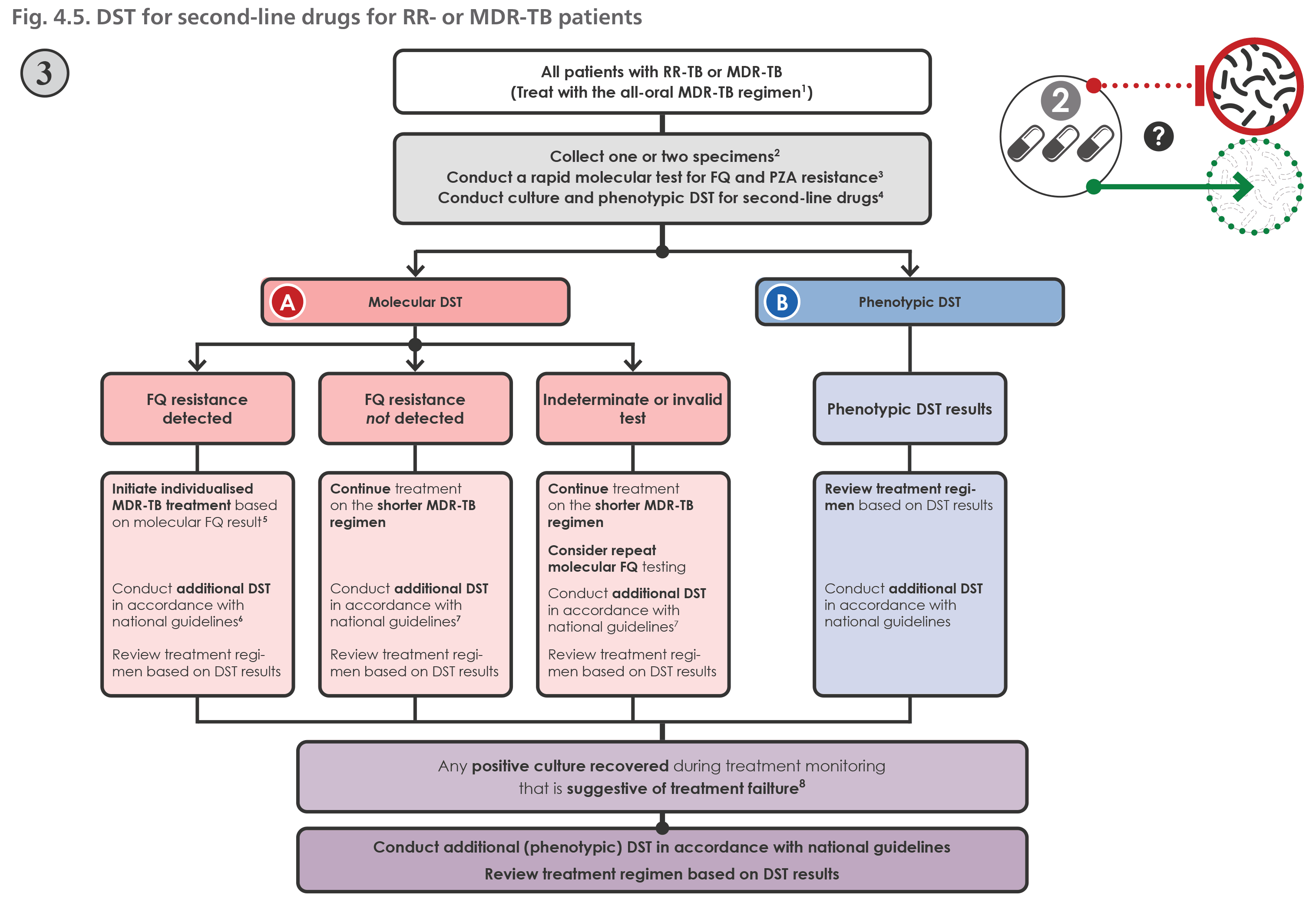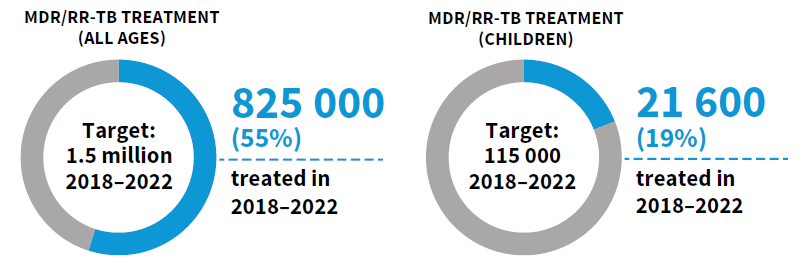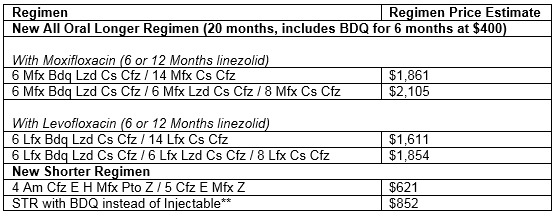
WHO's Rapid Communication on Treatment of Multidrug- and Rifampicin-Resistant Tuberculosis | Stop TB Partnership

MDR/XDR-TB management of patients and contacts: Challenges facing the new decade. The 2020 clinical update by the Global Tuberculosis Network - ScienceDirect
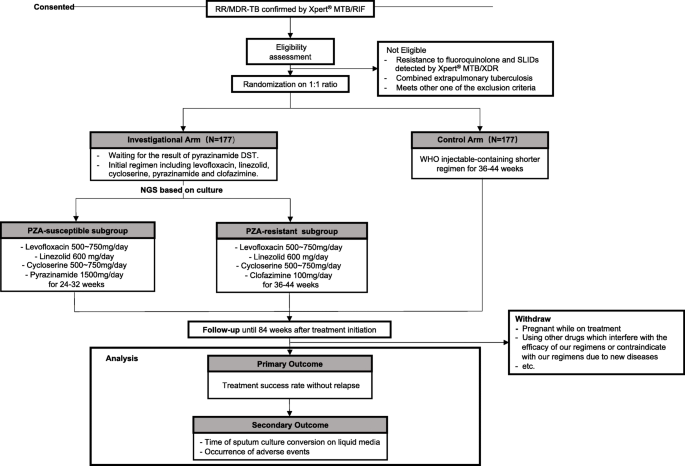
Refining MDR-TB treatment regimens for ultra short therapy (TB-TRUST): study protocol for a randomized controlled trial | BMC Infectious Diseases | Full Text

Keeping up with the guidelines: design changes to the STREAM stage 2 randomised controlled non-inferiority trial for rifampicin-resistant tuberculosis | Trials | Full Text

The global impact of household contact management for children on multidrug-resistant and rifampicin-resistant tuberculosis cases, deaths, and health-system costs in 2019: a modelling study - The Lancet Global Health

Drugs and doses used in shorter MDR regimen for patients older than 14... | Download Scientific Diagram
![PDF] WHO Treatment Guidelines for Drug-Resistant Tuberculosis, 2016 Update: Applicability in South Korea | Semantic Scholar PDF] WHO Treatment Guidelines for Drug-Resistant Tuberculosis, 2016 Update: Applicability in South Korea | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/06b1491afd422ac031fced965478fa59ddecfe52/4-Table2-1.png)
PDF] WHO Treatment Guidelines for Drug-Resistant Tuberculosis, 2016 Update: Applicability in South Korea | Semantic Scholar
Factors associated with treatment outcome of MDR/RR-TB patients treated with shorter injectable based regimen in West Java Indonesia | PLOS ONE

Selecting an appropriate all-oral short-course regimen for patients with multidrug-resistant or pre-extensive drug-resistant tuberculosis in China: A multicenter prospective cohort study - ScienceDirect
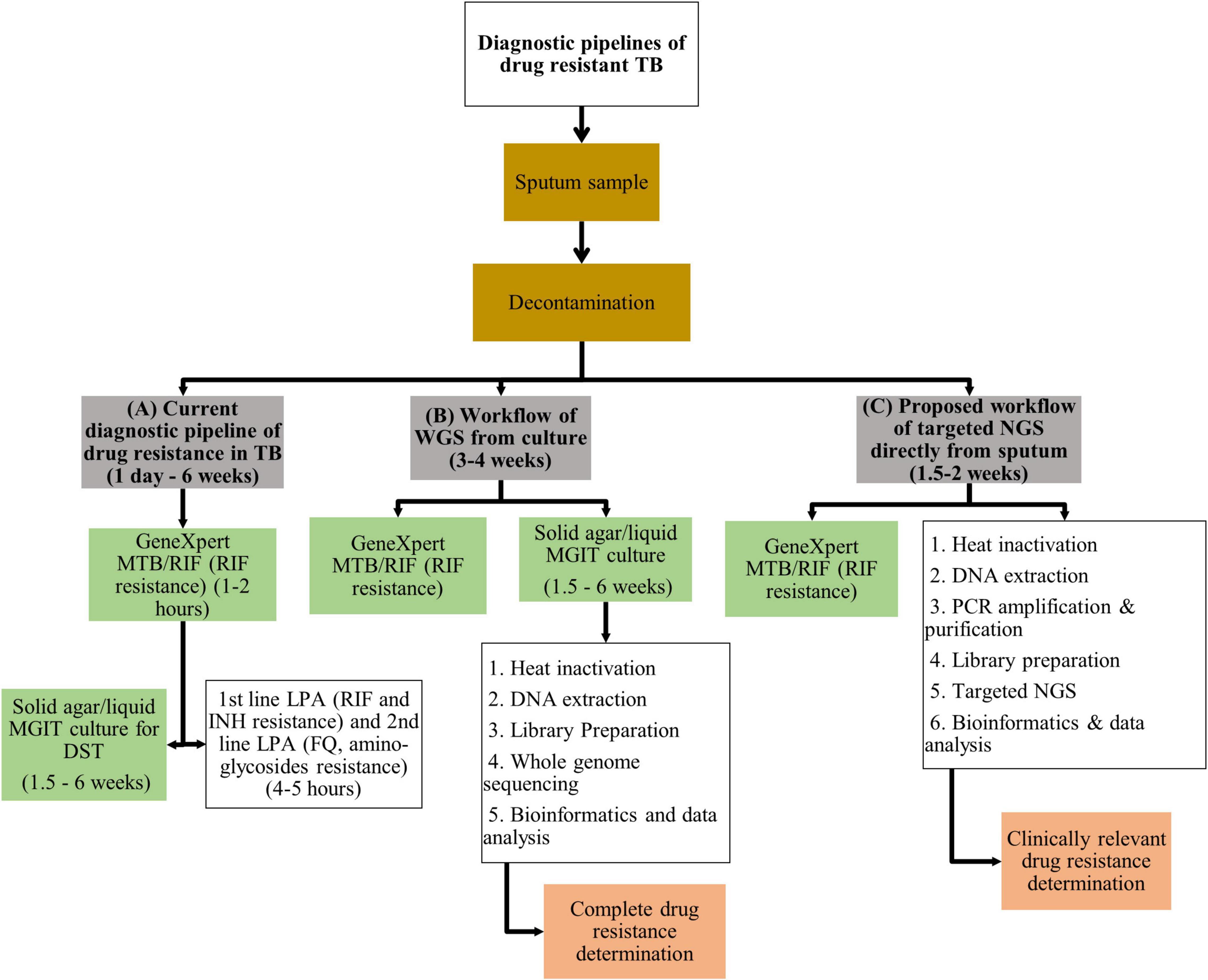
Frontiers | Application of Next Generation Sequencing for Diagnosis and Clinical Management of Drug-Resistant Tuberculosis: Updates on Recent Developments in the Field

Drug-associated adverse events in the treatment of multidrug-resistant tuberculosis: an individual patient data meta-analysis - The Lancet Respiratory Medicine

9 months of delamanid, linezolid, levofloxacin, and pyrazinamide versus conventional therapy for treatment of fluoroquinolone-sensitive multidrug-resistant tuberculosis (MDR-END): a multicentre, randomised, open-label phase 2/3 non-inferiority trial in ...